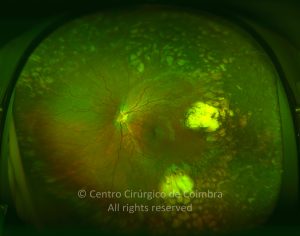
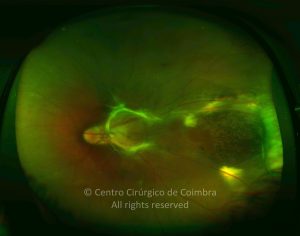
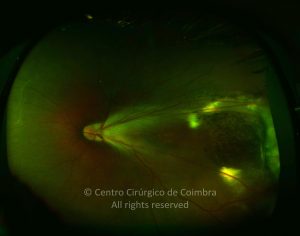
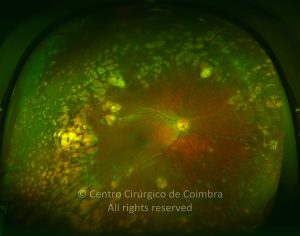
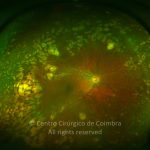
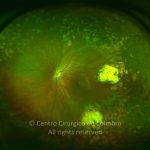
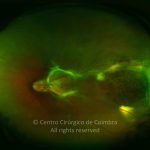
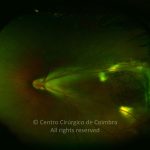
Retinopathy of prematurity (ROP) is an ischemic retinopathy of premature and low-birth-weight infants.
Vascularization of the retina starts by week 16 of gestation. It develops centrifugally from the optic disc towards the ora serrata. The nasal retina is usually vascularized by the 8th month of gestation, and the vascularization of the temporal ora is usually completed 1-2 months later. When a child is born prematurely with an incomplete retinal vascularization, ROP can develop. The risk factors associated with ROP include prematurity, low birth weight and supplemental oxygen in neonatal care.
Early treatment for retinopathy of prematurity study found a prevalence of ROP in children with birth weight 1000-1250g of 44%, 751-999g of 76% and << 750g of 93%. < Supplemental oxygen was also related to increased incidence of ROP. The first attempts to reduce supplemental oxygen in premature infants, resulted in higher mortalities. With the widespread use of pulse oximetry and systems allowing better control of oxygen delivered, a decreased incidence of ROP, without increased mortality and morbidity was possible.
Several classifications have been developed to stratify the patients with ROP. The major on used in clinical practice is the International Classification of ROP. We consider the details of this classification beyond the scope of this Atlas.
Early diagnosis is paramount. Fundus examination should be performed on infants born with gestational age of 30 weeks or less, a birth weight of less than <1500 g or a birth weight of 1500-2000 g when oxygen supplementation was required. The first examination should be done at 4 weeks to less than 5 weeks after birth or at a corrected gestational age of 30 to less than 31 weeks, whichever is later.
The standard treatment was based almost exclusively on laser photocoagulation of the avascular retina, an effective method for stabilizing the disease, but associated with tissue destruction and long-term changes in the visual field. Intravitreal injection of a reduced volume of anti-VEGF (0.03 ml) in premature infants demonstrated clinical efficacy and safety, establishing a new therapeutic approach for retinopathy of prematurity.